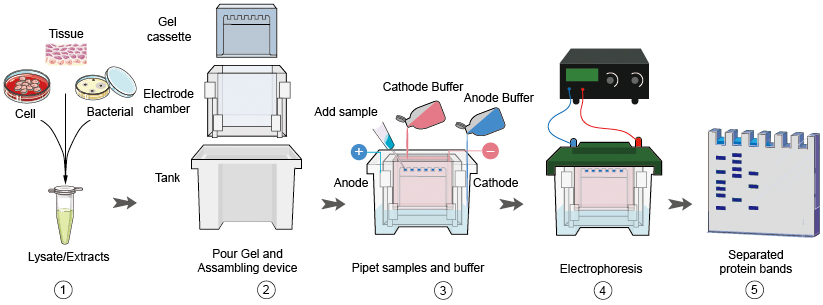
You should be able to see bubbles rising through the tank. Turn on the power supply and set the voltage recommended by the manufacturer of the gels in the gel tank. Once all the samples are loaded, add running buffer, place the lid onto the electrophoresis tank. All the samples contain equal amounts of protein. Load a molecular weight market into the first lane then load the samples into adjacent wells. Acrylamide percentage of the gel being used depends on the molecular weight of the target protein. Place a gel into the electrophoresis tank and add in buffer, ensuring the tops of the wells are covered. This results in linearized proteins with a negative charge proportional to their size. 
Heat the samples and 95 degrees C for five to 10 minutes in a sample buffer containing a reducing agent such as beta-mercaptoethanol. The proteins are then transferred onto a membrane where they can be detected using antibodies. The purpose of western blotting is to separate proteins on a gel according to the molecular weight. Protocols are provided by Abcam “AS-IS” based on experimentation in Abcam’s labs using Abcam’s reagents and products your results from using protocols outside of these conditions may vary. Watch our easy-to-follow video protocols.View AbExcel secondary antibodies for exceptional western blots.View our list of available positive control lysates, blocking peptides, and positive control proteins.Lysates can be aliquoted and stored at -20☌ for future use.Īll lanes: beta Actin antibody - loading control (ab8227) at 1/5000 dilution To reduce and denature your samples, boil each cell lysate in sample buffer at 100☌ for 5 min. 
We recommend reducing and denaturing the samples using the following method unless the online antibody datasheet indicates that non-reducing and non-denaturing conditions should be used.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
